THE NEXT GENERATION OF NASAL DRUG DELIVERY
INNOVATION IN TARGETING
Where Molecules Meet the Brain
Some molecules in vapor form—such as those in perfumes—are known to reach the brain via the olfactory nerve. However, delivering drugs in liquid or solid form to the upper nasal cavity, where this nerve is located, has been extremely challenging. Overcoming this barrier is essential for effective nose-to-brain drug delivery.
TACKLING THE BRAIN’S TOUGHEST BARRIER
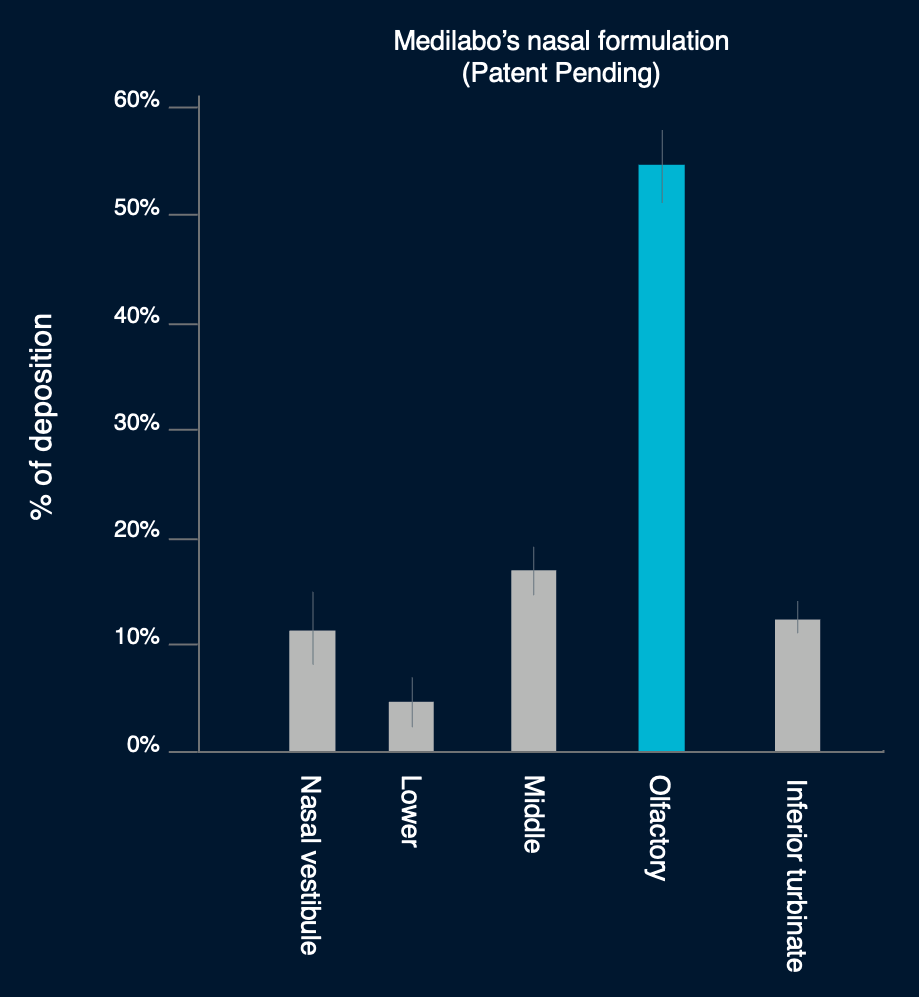
The Breakthrough: Targeting the Olfactory Zone
By optimizing the formulation, over 50% of the dose reaches the olfactory region—allowing direct brain access while avoiding the liver and bloodstream.
Key Benefits
・Bypasses first-pass metabolism for better brain delivery
・Reduces systemic exposure and side effect risk
・Enables CNS use of drugs unsuitable for oral delivery
Repurposing with Precision
We analyzed compounds with strong in vitro and in vivo effects that failed in clinical trials—often due to poor pharmacokinetics, not biology. From this, two standout candidates emerged: rifampicin and resveratrol. Both hold high potential for CNS treatment when delivered via our optimized intranasal platform.
OUR PIPELINE
| PROGRAM | INDICATION | DESCRIPTION | STAGE |
|---|---|---|---|
| ML1707 | •Alzheimer’s Disease •Frontotemporal Dementia | Nasal rifampicin, tau oligomer inhibitor ( Orphan Drug Designation ) | Preclinical |
| ML2404 | •Maternal depression •Major depression | Nasal resveratrol,restore mitochondrial function | Preclinical |
| ML 1808 | •Alzheimer’s Disease •Frontotemporal Dementia •Parkinson’s Disease | Nasal combination, rifampicin + resveratrol,higher efficacy and safety | Preclinical |
OUR TEAM

CEO
Co-Founder
Dr. Kumagai brings over two decades of experience in drug development and commercialization. At Astellas Pharma, he led programs across multiple therapeutic areas, contributing to four successful drug launches. He also spearheaded the market introduction of a novel gait assessment system at GE Healthcare. Holding a PhD in pharmaceutical sciences and an MBA from Globis University, his award-winning strategic vision focuses on de-risking CNS drug development through translational innovation.

CMO
Dr. Kuerner brings over 25 years of experience in clinical development and regulatory strategy across Japan, the U.S., and Europe. At Boehringer-Ingelheim Japan, he led new drug development and post-marketing programs throughout Asia. With a medical background in internal medicine and molecular biology, he has managed large multinational teams and driven cross-border collaboration, combining scientific expertise with strong leadership in global pharmaceutical development.

CSO
Co-Founder
Dr. Tomiyama is a leading neuroscientist known for discovering the Osaka mutation in familial Alzheimer’s disease and for his influential research on tau and amyloid-β oligomers. His work has advanced therapeutic strategies using rifampicin and deepened understanding of dementia pathogenesis. He specializes in translational research and currently leads the Translational Neuroscience Research division at Osaka Metropolitan University, bridging basic science and clinical application in neurodegenerative diseases.
NEWS
